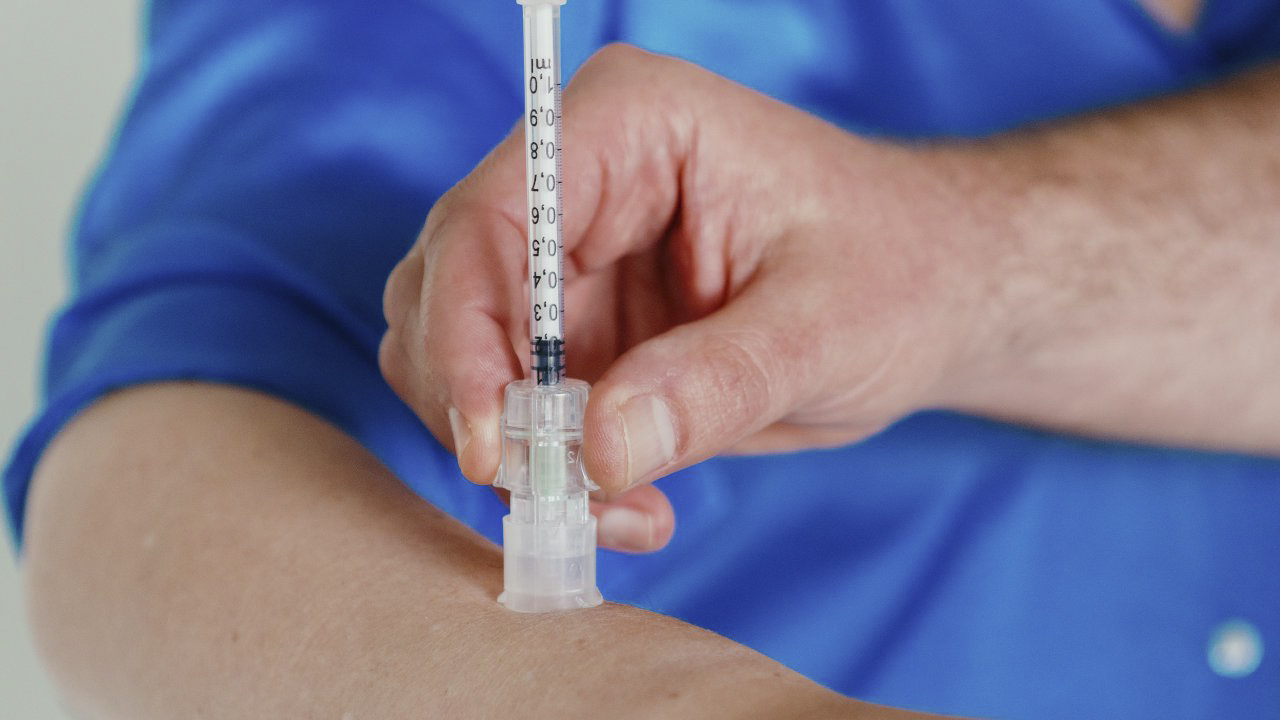
VAX-ID, a reliable patient-friendly intradermal injection adapter
VAX-ID® is the heart of Idevax, an ISO13485:2016 certified company. It is an award-winning patented intradermal injection adaptor suited for preventive, diagnostic and therapeutic applications.
VAX-ID® is CE marked as a Class IIa medical device, meeting the stringent European Medical Device Regulations for products sold within the European Economic Area (EEA). For other regions, devices are currently available for investigational use only (IUO) and research use only (RUO).
Watch how the device works
Why skin?
The skin is the body’s first line of defense, rich in immune cells and supported by a dense vascular and lymphatic network. This makes it an optimal—yet often overlooked—site for drug delivery.
Intradermal injection supports efficient immune responses while also supporting broader vaccine access, while also offering powerful applications across diagnostics, prevention, and advanced therapies.
Our Team:
We have a multidisciplinary and ambitious group. Our growing team consists of specialists in Research & Development, Quality Assurance, Regulatory Affairs, Manufacturing, Sales & Marketing. We also welcome several interns to our team every year. Additionally, our Idevax team is backed by a strong Scientific Advisory Board. Learn more about our team members here.