
Skin representation. Source: Layer by Layer: Understanding Your Skin’s Structure – Ask The Scientists [1].
Part 1: A look into the human skin
Physiology background
To understand skin drug delivery, first we need to get some knowledge about the skin itself. A basic principle in physiology, the branch of biology that studies the normal functions of living organisms and their parts, is that form and function are interconnected. That means, the form and characteristics of a certain cell, tissue or organ have the purpose of allowing the main function of that organ to be successfully performed. For instance, a muscle’s anatomy is evolved to be able to produce movement under the right conditions. Having this concept in mind helps to understand how the skin’s structure is. The skin is the largest organ in the human body and the first barrier against the outside world. Accordingly, this special organ developed all the characteristics necessary to successfully filter what goes in and out of our organism, hence protecting us against different external harms, including pathogens.
Basic skin histology and anatomy
The skin is a multifunctional organ part of the integumentary system with unique anatomy and cellular composition. It consists of layers functioning in a dynamic and interconnected way [2], see Figure 1:
– The epidermis, the outermost layer of the skin and therefore the layer in direct contact with the environment. Consequently, this layer behaves as a physical barrier and is not vascularized to avoid systemic entrance of undesired hazards. The epidermis is formed primarily by keratinocytes, a special cell type able to produce keratin that provides strength. This skin layer is divided in stratums and the outermost one, called stratum corneum is composed of a special cell type called corneocytes, interspersed in lipids, forming the so-called “brick and mortar model”. These are dead cells full of keratin which pileup over each other functioning as a “wall”. It also contains immune cells in case a pathogen breaches the barrier. Additionally, this layer hosts beneficial microbes that contribute to the skin’s protection against environmental threats.
– Then there is the underlying layer called dermis, mainly composed of connective tissue and characterized by abundant extracellular matrix (ECM), which supports the epidermis in nutrition and other aspects. That means, differently from the epidermis this layer is vascularized, therefore it could potentially represent an easy entrance door for microorganisms. That’s why it contains several types of immune cells in great numbers that will “guard” the location, identifying possible “invasions”. These immune cells are mainly located in the papillary dermis which is the upper region of the dermis.
– Finally, there is the hypodermis which is right under the dermis. This is also known as the subcutaneous tissue, which contains a lot of fat and connective tissue. It supports and drains the dermis through its large lymphatic vessels and veins.
Each layer has a different organization and composition that functions as a role to defend us from environmental hazards. Moreover, the skin contains appendages such as hair follicles, sweat and sebaceous glands which are important for accomplishing the many skin functions.
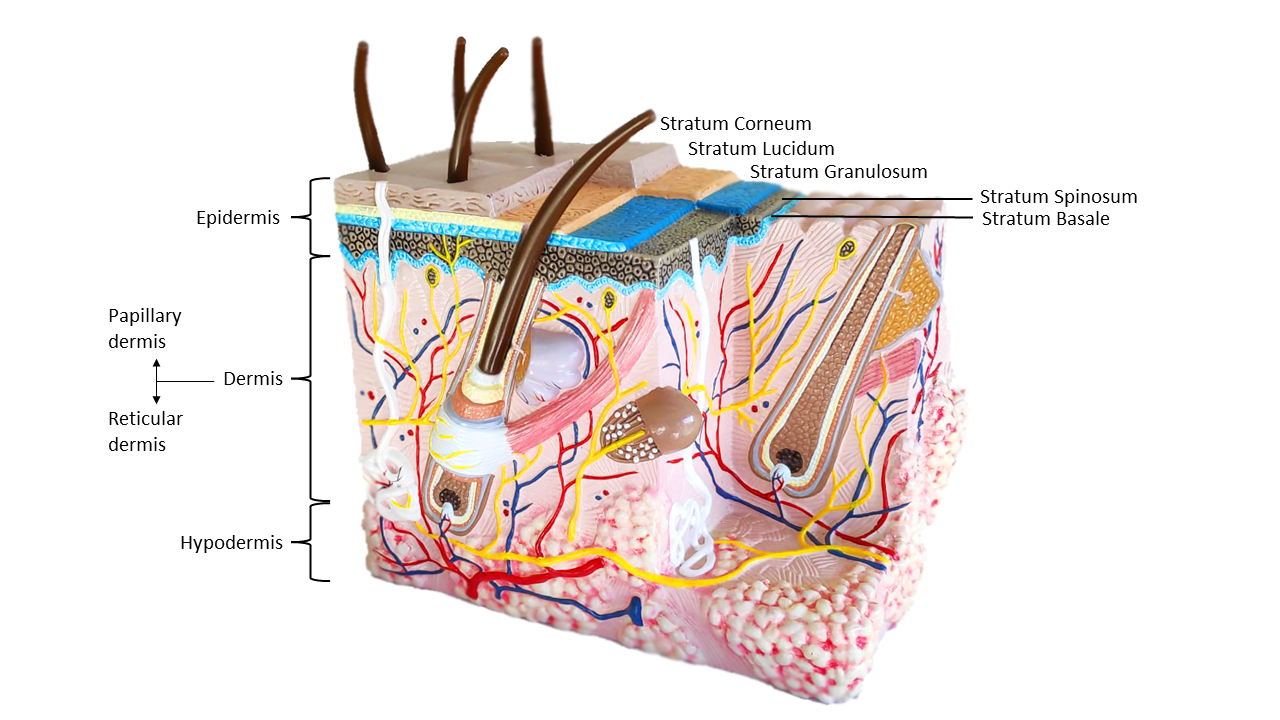
Figure 1 – Skin‘s layers. Source: IDEVAX.
Part 2: the immunological anatomy of the skin
Skin’s immune network
Besides the physical and chemical barriers promoted by the skin, this organ counts on a highly developed immune network that detects, and mounts coordinated immune responses against foreign invasions. These core barrier features of the skin provide selective entrance and distribution of molecules or microorganisms. Evidently, the skin immune network is the most important attribute for the administration of vaccines given that vaccines aim to generate protective immunogenicity against a certain antigen. The skin presents diverse immune-responsive components in great numbers, see Figure 2 [3], able to recognize antigens and trigger immune responses. There are several tissue-resident immune cells including antigen-presenting cells (APC) specific to the skin such as the Langerhans and dermal dendritic (DC) cells [4], macrophages, mast cells, dermal γδ T cells, and innate lymphoid cells [5]. The dermal DC cells capture and process antigens then migrate to locations rich in T-lymphocytes to initiate T-cell-mediated adaptive immune responses [6]. These immune cells interact with each other and with non-immune cells, such as peripheral nerves and the microbiota to orchestrate fine-tuned immune responses [7]. Accordingly, some strategies to improve the outcome of vaccinations target the skin DCs [8]. Even the skin appendages represent a unique immunological spot because it lacks the stratum corneum, which leaves the skin appendages in direct contact with the skin’s surface microbiota and environment which demands special attention on the immunological aspect. Furthermore, the rich ECM present in the skin dermis provides a scaffold for immune cell migration [7]. At this point, the physiology concept of how form and function are linked is clear, and it is possible to conclude that skin anatomy plays a key role in the dynamic behaviour of the skin immune network and therefore immune responses. On the contrary, when vaccines are delivered intramuscularly, the antigens bypass this skin immuno-surveillance activity and are passively drained via blood or lymph [9].
Consequently, the skin microenvironment can initiate both kinds of immunity [4] we have, the cellular, mediated by cells, and humoral, mediated by antibodies. Additionally, the skin can also trigger mucosal immunity which is particular to a few routes of administration (RoA). In fact, currently available vaccines elicit an inefficient cell-mediated immune response and lack protective mucosal immunity [10]. Therefore, skin vaccination can induce a more complete immune response than for example intramuscular (IM) vaccines. This is of special interest for pathogens that enter in contact with the host by mucosa (e.g. eyes, respiratory and gastro tracts), which by the way is the entrance door for the majority of all pathogens given that the mucosal surfaces are particularly vulnerable to infection [11].
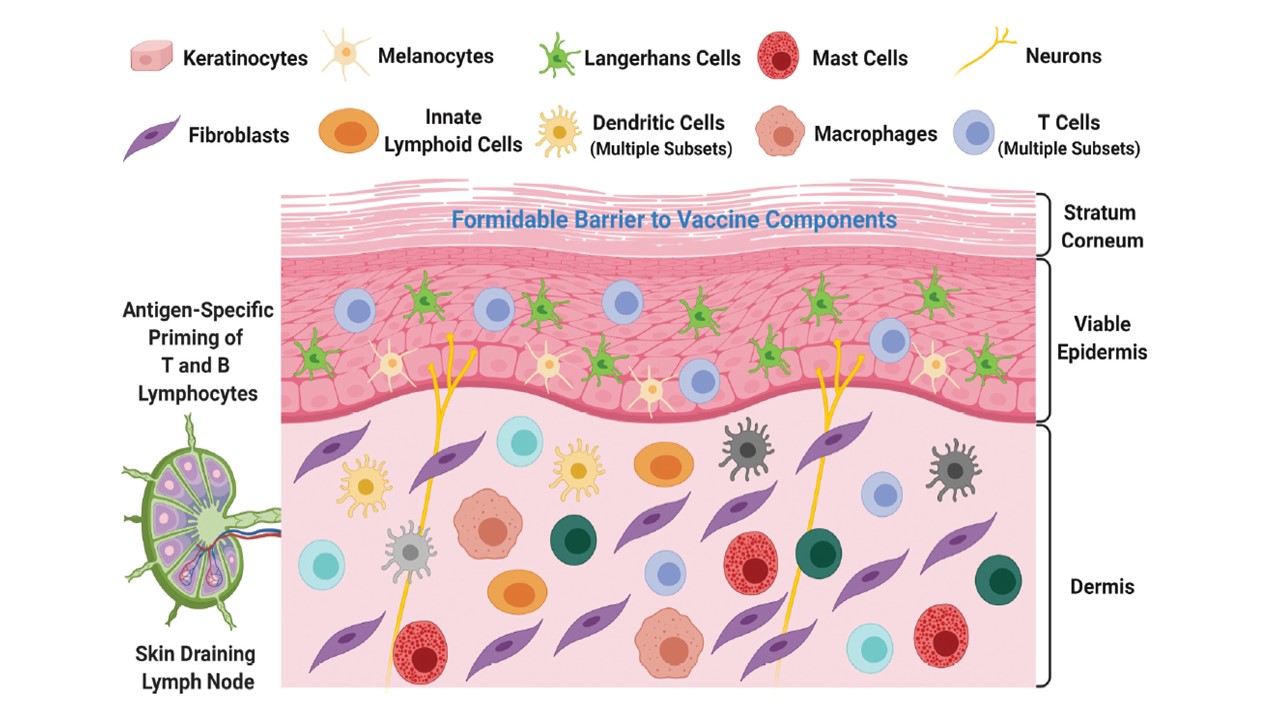
Figure 2 – Skin‘s immune-responsive components. Source: Korkmaz E. et al [2].
Advances in skin drug delivery
The intradermal delivery of vaccines represents a safe route and because it can limit the transfer of vaccine components to the blood circulation it may present a lower risk for septic shock or toxicity due to the hepatic first-pass effect [10]. Another benefit of skin drug delivery is that this route can be pain-free or incredibly low in pain which leads to enhanced patient compliance, especially in long-term therapies [10]. There is a variety of delivery strategies explored in this field that employ different skin drug delivery mechanisms. For instance, cutaneous drug delivery can rely on mechanisms such as the transcutaneous route and even the appendageal route.
Nevertheless, although the skin is a well-known optimal site for vaccination, the skin still is not the most common immunization site in clinical practice. This was linked to the lack of effective drug delivery systems that can enable safe, reproducible, and patient-friendly immunization. In response to this limitation, several new skin-targeted drug delivery strategies have emerged in the last years, such as sophisticated topical formulations, microneedle patches, needle adapters, etc [3]. These technologies explore different mechanisms for skin drug delivery depending on many factors. The continuous investigation of these platforms is likely to speed up the development of improved drug delivery techniques. Another factor pushing the advances in this field is the better understanding of the skin itself over the years. For instance, the recent advances in genomics, spectroscopy, and immunology have drastically contributed to a deeper understanding of the skin [12]. In conclusion, we can expect more drug delivery technologies to come which may contribute to greater adoption of skin drug delivery in clinical practice and research. Clearly, these are exciting times for researchers in the field.
References:
- Layer by Layer: Understanding Your Skin’s Structure – Ask The Scientists. <https://askthescientists.com/skin-layers/> Accessed on 03 March 2022.
- Junqueira J. C. Basic Histology: Text and Atlas-Text Only 13th edition. 2013.
- Korkmaz E. et al. Emerging skin-targeted drug delivery strategies to engineer immunity: A focus on infectious diseases. Expert Opin Drug Deliv. 2021 Feb;18(2):151-167. doi: 10.1080/17425247.2021.1823964. Epub 2020 Oct 6. PMID: 32924651.
- Lambert, P. H. & Laurent, P. E. Intradermal vaccine delivery: Will new delivery systems transform vaccine administration? Vaccine 26, 3197–3208 (2008).
- Tay SS. et al. Roediger B, Tong PL, Tikoo S, Weninger W. The Skin-Resident Immune Network. Curr Dermatol Rep. 2013 Nov 28;3(1):13-22. doi: 10.1007/s13671-013-0063-9. PMID: 24587975; PMCID: PMC3931970.
- Toebak, Mascha J., et al. “Dendritic cells: biology of the skin.” Contact dermatitis 60.1 (2009): 2-20.
- Kabashima K, Honda T, Ginhoux F, Egawa G. The immunological anatomy of the skin. Nat Rev Immunol. 2019 Jan;19(1):19-30. doi: 10.1038/s41577-018-0084-5. PMID: 30429578.
- Romani N. et al. (2011) Targeting Skin Dendritic Cells to Improve Intradermal Vaccination. In: Teunissen M. (eds) Intradermal Immunization. Current Topics in Microbiology and Immunology, vol 351. Springer, Berlin, Heidelberg. https://doi.org/10.1007/82_2010_118.
- Van Damme, P. et al. Safety and efficacy of a novel microneedle device for dose sparing intradermal influenza vaccination in healthy adults. Vaccine 27, 454–459 (2009).
- Criscuolo E et al. Alternative Methods of Vaccine Delivery: An Overview of Edible and Intradermal Vaccines. J Immunol Res. 2019 Mar 4;2019:8303648. doi: 10.1155/2019/8303648. PMID: 30949518; PMCID: PMC6425294.
- Immunobiology: The Immune System in Health and Disease. 5th edition. Janeway CA Jr, Travers P, Walport M, et al. New York: Garland Science; 2001.
- Gu Z, Chen X. Towards Enhancing Skin Drug Delivery. Adv Drug Deliv Rev. 2018 Mar 1;127:1-2. doi: 10.1016/j.addr.2018.05.004. PMID: 29903554.
Interested in our solutions?
Contact our commercial team!
